
Romane Maillet
Product Manager
Yeast and bacteria combination for the best of both worlds
Yeast and bacteria have been successfully used as probiotic for decades: bacteria, mainly from the Lactobacilli and Bifidobacteria species; and yeast Saccharomyces cerevisiae var. boulardii. The two microorganism’s act in two different mechanisms and therefore put forth complementary modes of action, which bring a dual action and additional benefits.
| Probiotic bacteria | Probiotic yeast – S. boulardii |
| Prokaryote cells | Unicellular eukaryote organism with cell wall |
| Shape: Coccus (spherical), bacillus (rod-shaped | Shape: Ovoid or spherical |
| Size: ~ 1 μm | Size: ~ 10 μm |
| Metabolism: Wide variety to survive extreme conditions | Metabolism: Lives in the presence or absence of O2 |
| Reproduction: By cell division every 20-30 minutes | Reproduction: By budding |
| Presence in the microflora: 99% | Presence in the microflora: < 1% |
| Sensitive to antibiotics | Sensitive to antifungal agents |
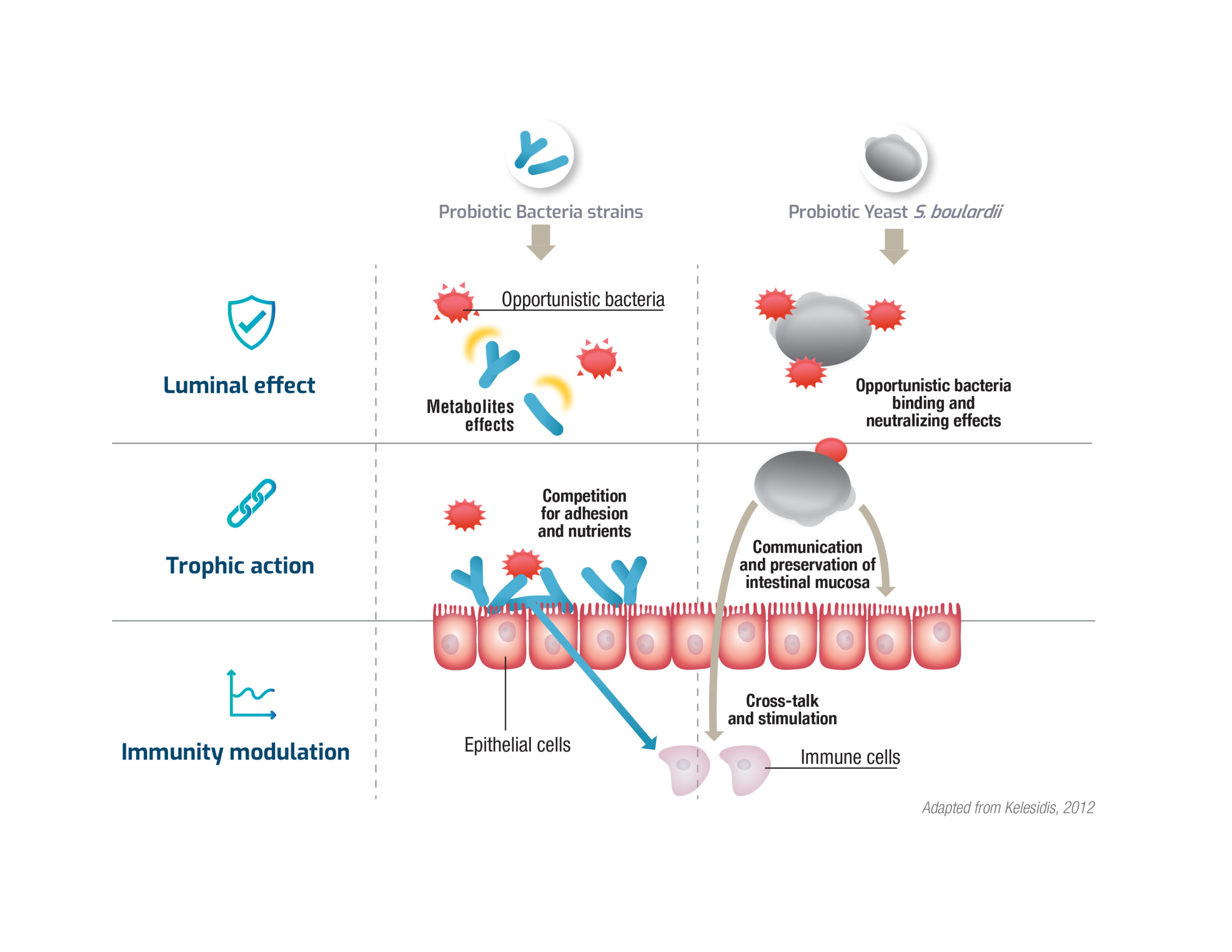
Complementary modes of action of probiotic bacteria and yeast for additional benefits
A core of science exposes the interaction, dual action, and proven benefit of combining yeast and bacteria. The two microorganism’s dissimilar functions and attributes put forth complementary modes of action. While yeast binds to opportunistic bacteria with its specific receptors on its cell wall, bacteria have an effect through the production of metabolites. Furthermore, looking at epithelial cells level, yeast communicate with epithelial cells as well as the intestinal microflora thus preserving barrier integrity and supporting a favorable environment for the microflora balance, whereas probiotic bacteria also compete with external bacteria for adhesion to epithelial cells and for nutrients. Finally, both yeast and bacteria can stimulate and modulate the immune system to help the body’s defenses.
Scientific Evidence
Harnessing the synergistic effect of S. boulardii CNCM I-1079 combined with bacteria
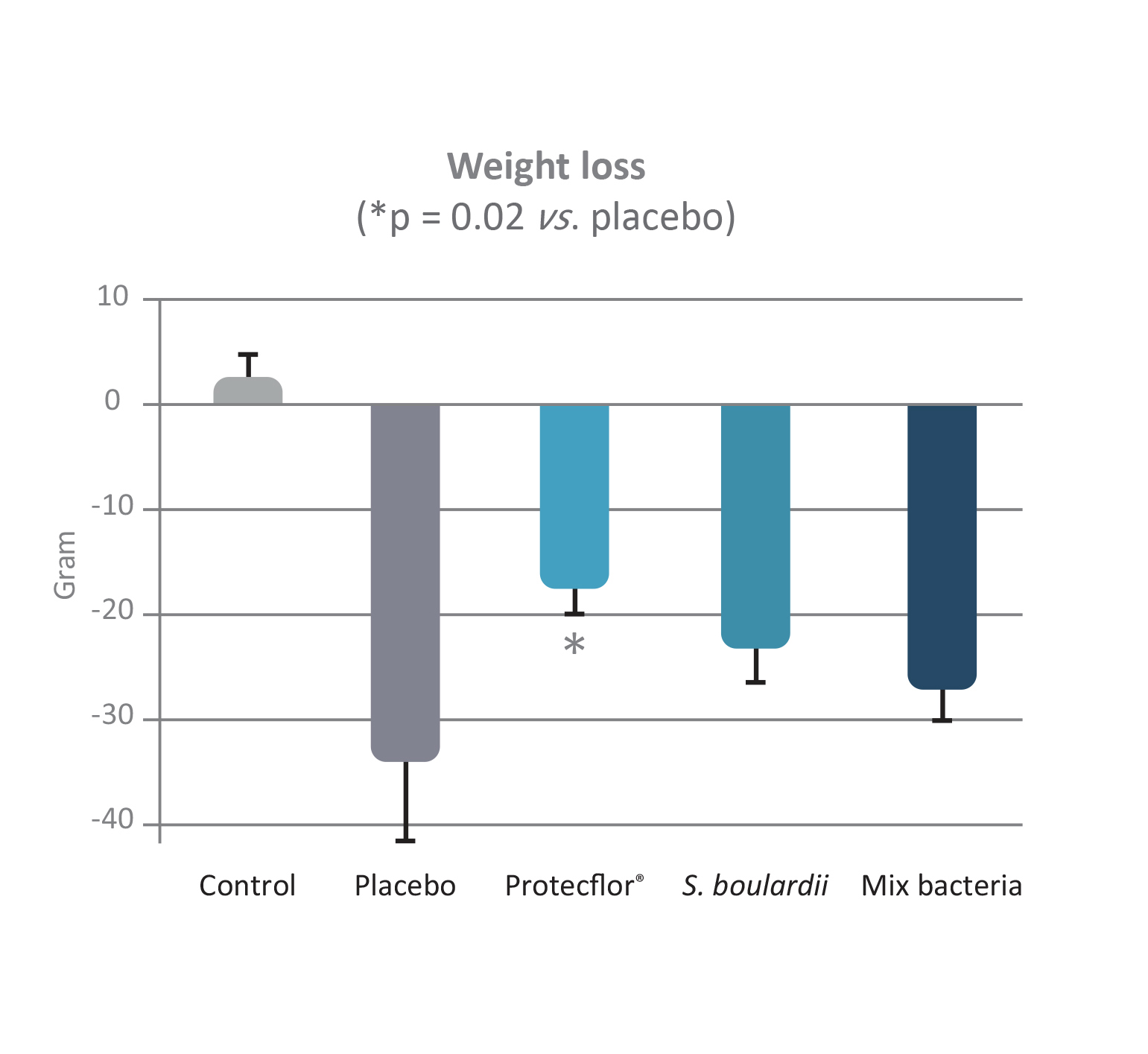
Protecflor® combination: S. boulardii CNCM I-1079, L. helveticus Rosell®-52, L. rhamnosus Rosell®-11 and B. longum Rosell®-175
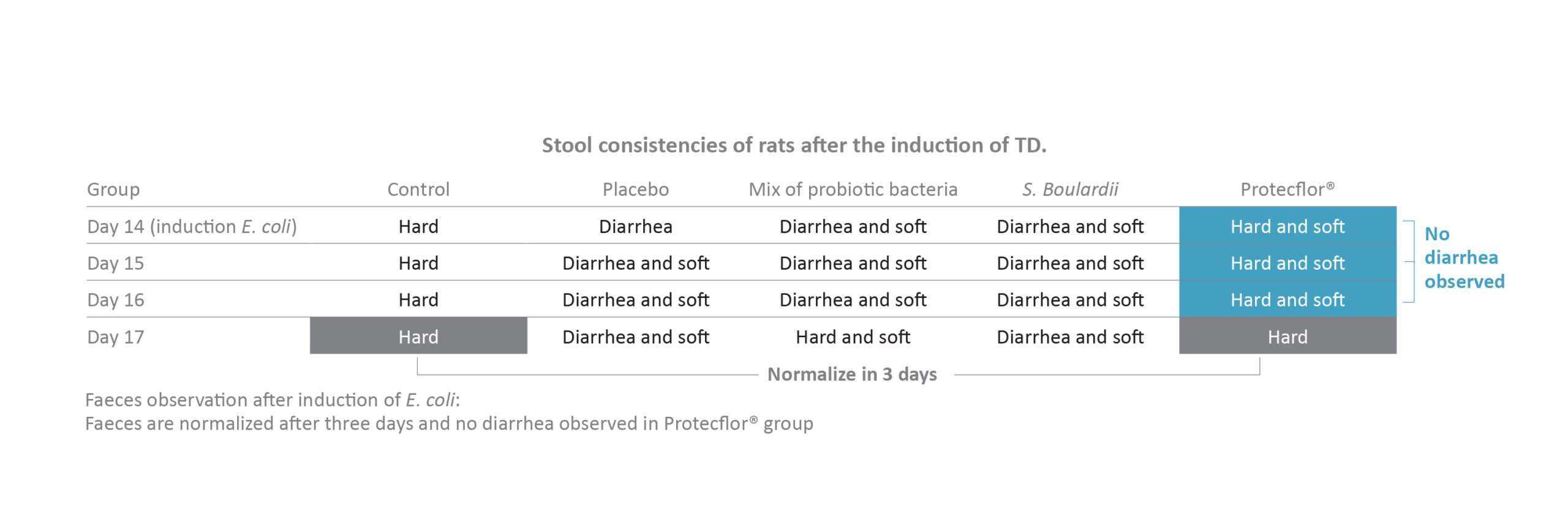
Protecflor® combination has been documented in two studies. In an in vivo model [Bisson 2009], the author evaluated the effect of the product on changes in the microbiome due to traveling induced by E. coli ETEC on rats. The infected rats were split into four groups administrated with S. boulardii CNCM I-1079 alone, mix of three probiotic bacteria, Protecflor® product or saline solution as placebo. A fifth group uninfected by E. coli ETEC was also used as control. Results showed a significantly greater effect with Protecflor® compared to placebo or S. boulardii alone or
the bacteria mix in several parameters including body weight loss (p = 0.02), body temperature (p <0.02) or modulation of inflammatory cytokine response (p <0.03). In addition, no diarrhea at all were observed in the Protecflor® group in this in vivo model.
 Grgov also confirmed the effectiveness of Protecflor® in a randomized controlled trial performed on 167 adults with the presence opportunistic microorganisms and administrated with a triple eradication therapy for seven days with or without probiotic combination for four weeks. Protecflor® significantly increased eradication rate after eight weeks compared to control (93.3% vs. 81.8%, p <0.05). The product also decreased the incidence of adverse effects vs. control (17.7% vs. 28.6%).
Grgov also confirmed the effectiveness of Protecflor® in a randomized controlled trial performed on 167 adults with the presence opportunistic microorganisms and administrated with a triple eradication therapy for seven days with or without probiotic combination for four weeks. Protecflor® significantly increased eradication rate after eight weeks compared to control (93.3% vs. 81.8%, p <0.05). The product also decreased the incidence of adverse effects vs. control (17.7% vs. 28.6%).

S. boulardii CNCM I-1079 and three Rosell® bacteria strains
Same bacteria strains as Protecflor® but different concentration and strain ratio.
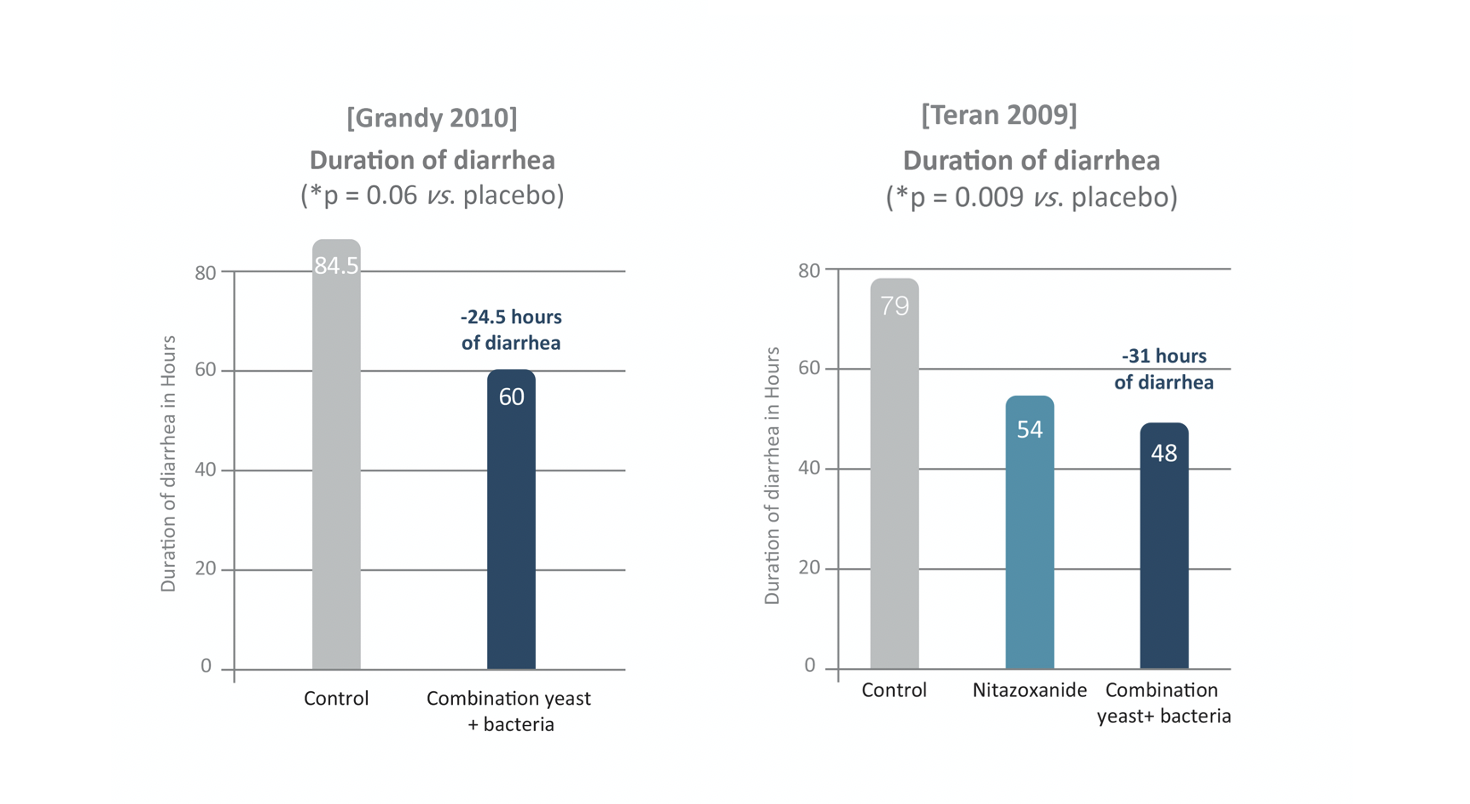
Two randomized controlled clinical studies were completed on infants with occasional diarrhea and also showed interesting results on yeast and bacteria combination. In the first one [Grandy 2010], 64 infants aged from one to 23-month old with diarrhea took oral rehydration solution (ORS) with or without probiotic combination twice daily for five days. In the second one [Teran 2009], the protocol included 75 infants from one to 24-month-old with the same health conditions split the same way, ORS with or without probiotics during same period.
Both assessed the efficiency of probiotic yeast and bacteria strains combination by revealing that the probiotic product acted on the significant reduction of vomiting duration compared to control (0 vs. 42.5 hours, p = 0.041) and the decrease of diarrhea duration (60 vs. 84.5 hours, p = 0.06) in the first trial [Grandy 2010]; and by significantly decreasing the duration of hospitalization (72 vs. 108 hours, p = 0.017) and diarrhea (48 vs. 79 hours, p = 0.009) compared to control in the second study [Teran 2009].
S. boulardii CNCM I-1079 and L. rhamnosus GG combination
In 2019, Moens evaluated the effect of the most documented probiotic yeast S. boulardii and the famous probiotic bacteria L. rhamnosus GG when combined together in an in vitro model simulating the human microbiota of the gastrointestinal tract after an infection with an opportunistic microorganism. After dysbiose induction of the microbiota due to an enterotoxigenic bacteria, 48 hours incubations were done with S. boulardii CNCM I-1079 alone or L. rhamnosus GG alone, or both probiotics in combination. Results exposed a significant greater decrease of toxin concentration as well as concentration of the opportunistic microorganism with S. boulardii in combination with L. rhamnosus GG compared to control.
Combination with other relevant ingredients
Lallemand Health Solutions continues its research to find new ingredients to combine with its probiotic products to confer additional effects. Therefore, Lallemand Health Solutions can provide a combination of S. boulardii with prebiotics including inulin and Fructooligosaccharides (FOS), or with minerals and vitamins. The company also works on the development of product containing the live microorganism yeast S. boulardii combined with derivatives yeast cell extract to provide yeast-based products.
Experts in Probiotic Formulation
Lallemand Health Solutions cumulates over 85 years of microbial fermentation production know-how to successfully combine probiotics bacteria, yeast and other health ingredients, such as botanical extracts, prebiotics, vitamins, and minerals. Every new ingredient must follow a strict validation process to ensure product compatibility, stability, and shelf-life longevity, following to ICH guidelines. From lab to shelf, Lallemand Health Solutions has the full control and expertise to produce premium tailored-made probiotic formulations in several galenic formats and packaging, in state-of-the-art facilities certified for the highest quality.
Contact:
lallemand-health-solutions.com
healthsolutions@lallemand.com
