Synthetic Biologics First Quarter 2019 Operational Highlights and Financial Results
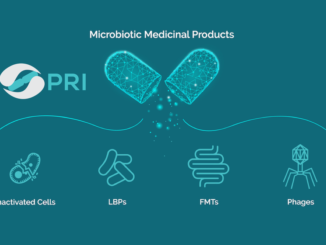
Synthetic Biologics, Inc. (NYSE American: SYN), a late-stage clinical company developing therapeutics that preserve the microbiome to protect and restore the health of patients, today provided an operational update and reported financial results for the […]